Glycogen balance
Animal glycogen, like amilopectin in plants, is
a branched homopolymer of glucose.
The glucose residues are linked by an α1à4-glucosydic bond. Every tenth or so glucose
residue has an additional α1à6 bond to another glucose. These branches are
extended by additional α1à4-linked glucose residues. This structure
produces tree-shaped molecule structure that consists of up to 50,000 residues.
Hepatyc glycogen is never completely degraded.
In general, only the non-reducing ends of the “tree” are shortened, or – when
glucose is abundant – elongated. The reducing end of the tree is linked to a
special protein, glycogenin. Glycogenin
carries out autocatalytic covalent bonding of the first glucose at one of its
tyrosine residues and elongation of this by up to seven additional glucose
residues. It is only at this point that glycogen
synthase becomes active to supply further elongation.
1)
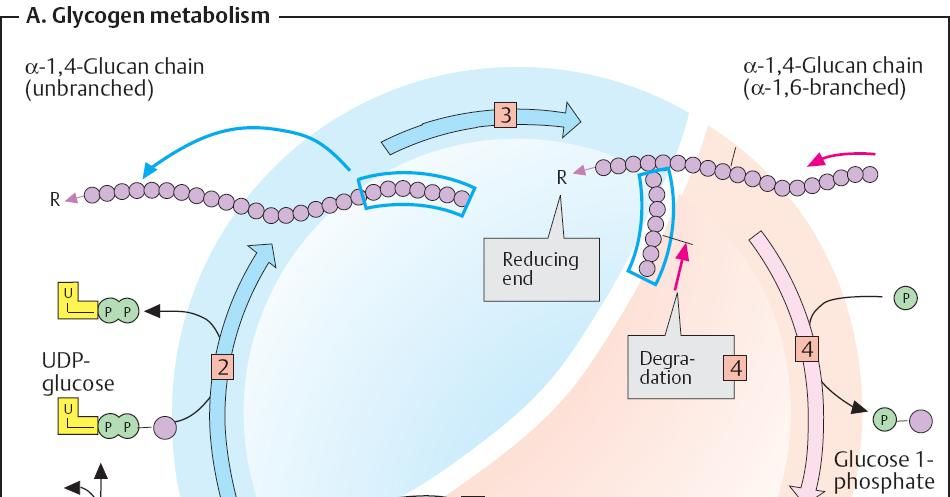
The
formation of glycosidic bonds between sugars is endergonic.
Initially, therefore, the activated form-UDP-glucose
– is synthesized by reaction of glucose
1-phosphate with UTP.
2)
Glycogen synthase now transfers glucose residues one by one from
UDP-glucose to the non-reducing ends of the available “branches”.
3)
Once the
growing chain has reached a specific length(>11 residues), the branching enzyme cleaves an
oligosaccharide, consisting of 6-7 residues from the end of it, and adds this
into the interior of the same chain or a neighboring one with α1à6 linkage. These branches are
then further extended by glycogen synthase.
4)
The
branched structure of glycogen allows rapid release of sugar residues. The most
important degradative enzyme, glycogen
phosphorylase, cleaves residues from a non-reducing end one after another
as glucose 1-phosphate. The larger
the number of these ends, the more phosphorylase molecules can attack
simultaneously. The formation of glucose 1-phosphate instead of glucose has the
advantage that no ATP is needed to channel the released residues into the
glycolysis or the PPP.
5)
Due to the
structure of glycogen phosphorylase, degradation comes to a halt four residues
away from each branching point. Two more enzymes overcome this point. First, a glucanotransferase moves a trisaccharide
from the side chain to the end of the main chain. A 1,6-glucosidase then cleaves the single remaining residues as a
free glucose and leaves behind an unbreached chain that is once again
accessible to phosphorylase.
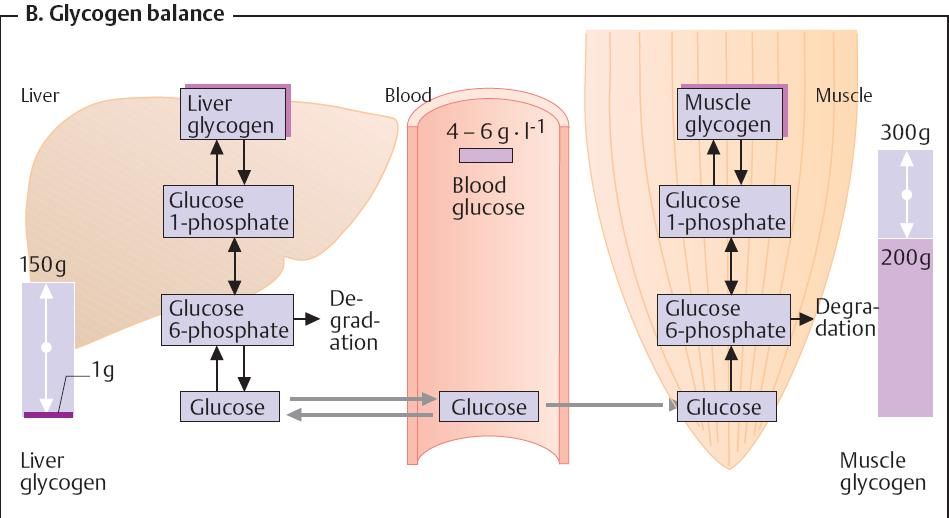
Glycogen balance
The human organism can store up to 450 g of
glycogen – one-third in the liver
and almost of the remainder in muscle.
The glycogen content of the other organs is low.
Hepatic
glycogen is mainly used
to maintain the blood glucose level in
the postresorptive phase. The
glycogen content of the liver therefore varies widely, and can decline to
almost zero in periods of extended hunger. After that, gluconeogenesis takes
over the supply for the organism. Muscle
glycogen serves as an energy
reserve and is not involved in blood glucose regulation. Muscle does
not contain any glucose-6-phosphatase and is therefore unable to release
glucose into the blood. The glycogen content of the muscle therefore does not
fluctuate as widely as that of the liver.
“Coloured atlas of biochemistry”, second edition; J. Koolman, K.H. Roehm









0 коментара:
Постави коментар