Our previous discussions of the psychological
responses to exercise have all been based on the conditions that exist at or
near sea level, where the barometric(air)
pressure(Pb) averages
about 760mmHg. Barometric pressure is a measure of the total pressure that all
of the gases comprising the atmosphere exert on the body(and everything else).
Regardless of the Pb, oxygen molecules always make up 20.93% of the
air. The partial pressure of oxygen(PO2) is
that portion of Pb exerted only by the oxygen molecules in the air.
At sea level, PO2 is therefore 0.2093 times 760mmHg, or 159mmHg.
Partial pressure is an important concept in understanding altitude physiology,
because it is primarily the low PO2 at altitude that limits exercise
performance. Although the human body tolerates small fluctuations in pressure,
large variations pose special problems. This is evident when mountain climbers
ascend to higher altitudes where reduced pressures can substantially impair
physical performance and can even jeopardize life.
The reduced barometric pressure at altitude is
referred to as a hypobaric
environment or simply hypobaria(low atmospheric pressure). The lower
atmospheric pressure also means a lower PO2, which limits pulmonary diffusion of oxygen from the
lungs and oxygen transport to the tissues. When oxygen delivery to the body
tissues is compromised, the result is cellular hypoxia(oxygen deficiency).
Hypobaric environments: conditions at altitude
Clinical problems associated with altitude were
reported as early as 400BC. However, most of the early concerns about ascent to
high altitudes focused on the cold conditions at altitude rather than the
limitations imposed by low air pressure. The initial landmark discoveries that
led to our current understanding of the reduced Pb and PO2
at altitude can be credited primarily to three 15th and 16th
century scientists. Torricelli(ca. 1644) developed the mercury barometer, an
instrument that permits the accurate measurement of atmospheric pressure. Only
a few years later(1648), Pascal demonstrated a reduction in barometric pressure
at high altitudes. Nearly 130 years after that(1777), Lavoisier described
oxygen and the other gases that contribute to the total barometric pressure.
The deleterious effects of high altitude on
humans that are caused by low PO2(hypoxia) were recognized in the
late 1800s. More recently, a team of scientists led by the late John Sutton
performed an intricate series of laboratory studies in the hypobaric chamber at
the U.S. Army Institute of Environmental Medicine. These experiments, known
collectively as Operation Everest II, have significantly added to our
understanding of exercise at altitude. For our discussion, the term altitude
refers to elevations above 1,500m(4,921ft), because few negative physiological
effects on performance are seen below that altitude.
While the major impact of altitude on exercise
physiology is attributable to the low PO2 that ultimatively limits
oxygen availability to the tissues, the atmosphere at altitude also differs in
other ways from sea-level conditions.
Atmospheric pressure at altitude
Air has weight. The barometric pressure at any
place on Earth is related to the weight of the air in the atmosphere above that
point. At sea level, for example, the air extending to the outermost reaches of
the earth’s atmosphere(approximately 38.6km, or 24mi) exerts a pressure equal
to 760mmHg. At the summit of Mount Everest, the highest point on Earth(8,848m,
or 29,028ft), the pressure exerted by the air above is only about 250mmHg. These
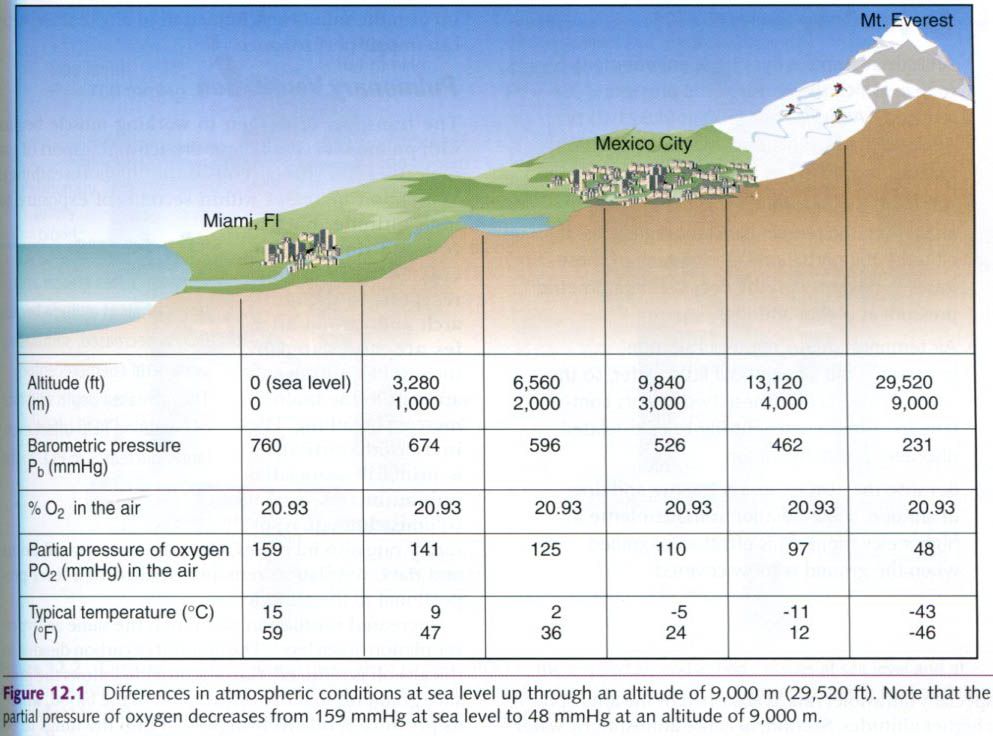
and related altitude differences are depicted in the picture below.
The barometric pressure on Earth does not
remain constant. Rather, it varies with changes in climatic conditions, time of
year, and the specific site at which the measurement is taken. On Mount Everest , for example, the mean barometric pressure
varies from 243mmHg in January to nearly 255mmHg in June and July. Also, the
earth’s atmosphere bulges outward slightly at the equator, which increases the
barometric pressure there by a few millimeters of mercury above standard
pressure. These variations are of little interest to people living near sea
level but are of considerable physiological importance for a climber attempting
to ascend Mount Everest without supplemental
oxygen.
Although barometric pressure varies, the
percentages of gases in the air that we breathe remain unchanged from sea level
to high altitude. At any elevation, the air always contains 20.93% oxygen,
0.03% carbon dioxide, and 79.04% nitrogen. Only the partial pressures change.
As shown in the figure above, the pressure that oxygen molecules in the air
exert at various altitudes drops proportionally with decreases in the
barometric pressure. The consequent changes in PO2 have significant effects on
the partial pressure of oxygen that reaches the lungs, as well as the gradients
between the alveoli of the lungs and the
blood(where oxygen is loaded) and between the blood and the tissues(where oxygen is unloaded).
Air temperature at altitude
Clearly, the low PO2 at altitude has
the greatest impact on exercise physiology. However, other environmental
factors contribute as well. For example, air temperature decreases at a rate of
about 1°C(1.8°F) for every 150m(about 490ft) of ascent. The average temperature
near the summit of Mount Everest is estimated
to be about -40°C(-40°F), whereas the sea level the temperature would be about
15°C(59°F). The combination of low temperatures, low absolute humidity, and
high winds at altitude poses a serious risk of cold-related disorders, such as hypothermia and windchill injuries.
Because of the cold temperatures at altitude,
the absolute humidity is extremely low. Cold air holds very little water. Thus,
even if air is fully saturated with water(100% relative humidity), the actual
amount of water contained in the air is small. The partial pressure of water,
also known as the water vapor pressure(PH2O), at 20°C(68°F) is about
17mmHg. But at -20°C(-4°F), this pressure decreases to only about 1mmHg. The
very low PH2O at high altitude promotes evaporation of moisture from
the skin(or clothing) surface, because of the high gradient between skin and
air, and can lead quickly to dehydration. In addition, a large volume of water
is lost through respiratory evaporation attributable to the dry air and increased
respiration rate experienced at altitude.
Solar radiation at altitude
The intensity of solar radiation increases at
high altitude for two reasons. First, at high altitudes, light travels through
less of the atmosphere before reaching the earth. For this reason, less of the
sun’s radiation, especially ultraviolet rays, is absorbed by the atmosphere at
higher altitudes. Second, because atmospheric water normally absorbs a
substantial amount of the sun’s radiation, the low water vapor in the air at
altitude also increases radiant exposure. Solar radiation may be further
amplified by reflective light from snow, which is usually found at higher
elevations.









3 коментара:
AMI high performance Oxygen Analyzers, O2 Sensors, Monitor, and Deficiency are guarantee to be higher quality and exceptional service at a lower price.
Moisture Analyzers
solar light pole
Seo executive near me
Постави коментар