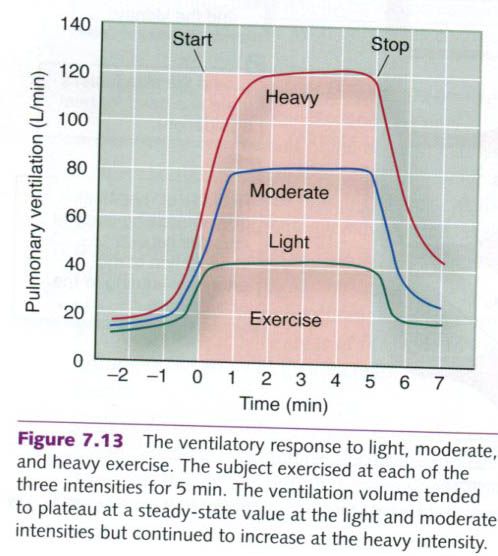
The onset of exercise is accompanied by an
immediate increase in ventilation. In fact, like the HR response, the marked
increase in breathing may occur even before the onset of muscular contractions.
This is shown in the picture below for light, moderate and heavy exercise.
Because of its rapid onset, this initial respiratory adjustment to the demands
of exercise is undoubtedly neural in nature, mediated by respiratory control
centers in the brain(central command), although neural input can also come from
receptors in the exercising muscle.
The more gradual second phase of the
respiratory increase shown in the picture is controlled primarily by changes in
the chemical status of the arterial blood. As exercise progresses, increased
metabolism in the muscles generates more carbon dioxide and H+.
Recall that these changes enhance oxygen unloading in the muscles, which
increases the (a-ṽ)O2 difference. Increased CO2 and H+ are
sensed by chemoreceptors primarily located in the brain, carotid bodies, and
lungs, which in turn stimulate the inspiratory center, increasing rate and
depth of respiration. Some researchers have suggested that chemoreceptors in
the muscles might also be involved. In addition, data suggest that receptors in
the right ventricle of the heart send information to the inspiratory center so
that increases in cardiac output can stimulate breathing during the early minutes
of exercise. These humoral influences on breathing rate and pattern serve to
fine-tune the respiratory response to exercise so as to match oxygen delivery
with aerobic demands without overtaxing respiratory muscles.
At the end of exercise, the muscles’ energy
demands decrease almost immediately to resting levels. But pulmonary
ventilation returns to normal at a slower rate. If the rate of breathing
perfectly matched the metabolic demands of the tissues, respiration would
decrease to the resting level within seconds after exercise. But respiratory
recovery takes several minutes, which suggests that postexercise breathing is
regulated primarily by acid-base balance, the partial pressure of dissolved
carbon dioxide(PCO2), and blood temperature.
Breathing irregularities during exercise
Ideally, breathing during exercise is regulated
in a way that maximizes aerobic performance. However, respiratory dysfunction
during exercise can hinder performance.
Dyspnea
The sensation of dyspnea(shortness of breath) during exercise is common among
individuals in poor physical condition who attempt to exercise at levels that
significantly elevate arterial carbon dioxide and H+ concentrations.
Both stimuli send strong signals to the inspiratory center to increase the rate
and depth of ventilation. Although exercise – induced dyspnea is sensed as an
inability to breathe, the underlying cause is an inability to adjust breathing
to blood PCO2 and H+.
Failure to reduce these stimuli during exercise
appears to be related to poor conditioning of respiratory muscles. Despite a
strong neural drive to ventilate the lungs, the respiratory muscles fatique
easily and are unable to reestablish normal homeostasis.
Hyperventilation
The anticipation of or anxiety about exercise,
as well as some respiratory disorders, can cause an increase in ventilation in
excess of that needed for exercise metabolism. Such overbreathing is termed hyperventilation. At rest,
hyperventilation can decrease the normal PCO2 of 40mmHg in the
alveoli and arterial blood to about 15mmHg. As arterial carbon dioxide
concentrations decrease, blood pH increases. These effects combine to reduce
the ventilatory drive. Because the blood leaving the lungs is nearly always
about 98% saturated with oxygen, an increase in the alveolar PO2
does not increase the oxygen content of the blood. Consequently, the reduced
drive to breathe – along with the improved ability to hold one’s breath after
hyperventilating – results from carbon dioxide unloading rather than increased
blood oxygen. Even when performed for only a few seconds, such deep, rapid
breathing can lead to light-headedness and even loss of consciousness. This
phenomenon reveals the sensitivity of the respiratory system’s regulation of
carbon dioxide and pH.
Valsalva
maneuver
The Valsalva
maneuver is a potentially dangerous respiratory procedure that frequently
accompanies certain types of exercise, in particular the lifting of heavy
objects. This occurs when the individual:
- Closes the glottis(the
opening between the vocal cords);
- Increases the
intra-abdominal pressure by forcibly contracting the diaphragm and the
abdominal muscles,
- Increases the intrathoracic
pressure by forcibly contracting the respiratory muscles.
As a result of these actions, air is trapped
and pressurized in the lungs. The high intra-abdominal and intra-thoracic
pressures restrict venous return by collapsing the great veins. This maneuver,
if held for an extended period of time, can greatly reduce the volume of blood
returning to the heart, decreasing cardiac output and altering arterial blood pressure. Although the Valsalva maneuver can be helpful
in certain circumstances, this maneuver can be dangerous and should be avoided.
Ventilation and energy metabolism
During long periods of mild steady-state
activity, ventilation matches the rate of energy metabolism, varying in
proportion to the volume of oxygen consumed and the volume of carbon dioxide
produced(VO2 and VCO2 respectively) by the body.
Ventilatory
equivalent for oxygen
The ratio between the volume of air expired or
ventilated(Ve) and the amount of oxygen consumed by the tissues(VO2)
in a given amount of time is referred to as the ventilatory
equivalent for oxygen, or Ve/VO2. It is typically measured in
liters of air breathed per liter of oxygen consumed per minute.
At rest, the Ve/VO2 can range from
23 to 28L of air per liter of oxygen. This value changes very little during
mild exercise, such as walking. But when exercise intensity increases to
near-maximal levels, the Ve/VO2 can be greater than 30L of air per
liter of oxygen consumed. In general, however, the Ve/VO2 remains
relatively constant over a wide range of exercise intensities, indicating that
the control of breathing is properly matched to the body’s demand of oxygen.
Ventilatory
threshold
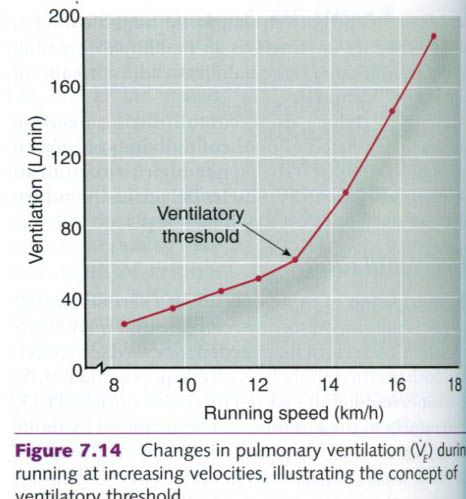
As exercise intensity increases, at some point
ventilation increases disproportionately to oxygen consumption. The point at
which this occurs is called the ventilatory
threshold, illustrated in
the figure below. When work rate exceeds ~55% to 70% of VO2max, at
approximately the same point as the ventilatory threshold, more lactate starts
to appear in the blood. This may result from greater production of lactate or
less clearance of lactate. This lactic acid combines with sodium bicarbonate(which
buffers acid) and forms sodium lactate, water, and carbon dioxide. As it is
known, the increase in carbon dioxide stimulates chemoreceptors that signal the
inspiratory center to increase ventilation. Thus, the ventilatory threshold
reflects the respiratory response to increased carbon dioxide levels.
Ventilation increases dramatically beyond the ventilatory threshold.
Respiratory
limitations to performance
Like all tissue activity, respiration requires
energy. Most of this energy is used by the respiratory muscles during pulmonary
ventilation. At rest, the respiratory muscles account for only about 2% of the
total oxygen uptake. As the rate and depth of ventilation increase, so does the
energy cost of respiration. The diaphragm, the intercostal muscles, and the
abdominal muscles can account for up 11% of the total oxygen consumed during
heavy exercise and can receive up to 15% of the cardiac output. During recovery
from dynamic exercise, sustained elevations in ventilation continue to demand
increased energy, accounting for 9% to 12% of the total oxygen consumed
postexercise.
Although the muscles of respiration are heavily
taxed during exercise, ventilation is sufficient to prevent an increase in
alveolar PCO2 or a decline in alveolar PO2 during activities
lasting only a few minutes. Even during maximal effort, ventilation usually is
not pushed to its maximal capacity to voluntarily move air in and out of the
lungs. This capacity is called the maximal voluntary ventilation and is
significantly greater than ventilation at maximal exercise. However,
considerable evidence suggests that pulmonary ventilation might be a limiting
factor during very high intensity(95~100% VO2max) exercise in highly
trained subjects.
Can heavy breathing for several hours(such as
during marathon running) cause glycogen depletion and fatique of the respiratory muscles? Animal studies have shown
a substantial sparing of their respiratory muscle glycogen compared with muscle glycogen in exercising muscles.
Although similar data are not available for humans, our respiratory muscles are
better designed for long-term activity than are the muscles in our extremities.
The diaphragm, for example, has two to three times more oxidative
capacity(oxidative enzymes and mitochondria) and capillary density than other
skeletal muscle. Consequently, the diaphragm can obtain more energy from
oxidative sources than can skeletal muscles.
Similarly, airway resistance and gas diffusion
in the lungs do not limit exercise in a normal, healthy individual. The volume
of air inspired can increase 20- to 40-fold with exercise – from ~5L/min at
rest up to 100 to 200 L/min with maximal exertion. Airway resistance, however,
is maintained at near-resting levels by airway dilation(through an increase in
the laryngeal aperture and bronchodilation). During submaximal and maximal
efforts in untrained and moderately trained individuals, blood leaving the
lungs remains nearly saturated with oxygen(~98%). However, with maximal
exercise in some highly trained elite endurance athletes, there is too large a
demand on lung gas exchange, resulting in a decline in arterial PO2
and arterial oxygen saturation(i.e. exercise-induced arterial hypoxemia, EIAH).
Approximately 40% to 50% of elite endurance athletes experience a significant
reduction in arterial oxygenation during exercise approaching exhaustion.
Arterial hypoxemia at maximal exercise is likely the result of a mismatch
between ventilation and perfusion of the lung. Since cardiac output is
extremely high in elite athletes, blood is flowing through the lungs at a high
rate and thus there may not be sufficient time for that blood to become
saturated with oxygen. Thus, in healthy individuals, the respiratory system is
well designed to accommodate the demands of heavy breathing during short- and
long-term physical effort. However, some highly trained individuals who consume
unusually large amounts of oxygen during exhaustive exercise can face
respiratory limitations.
The respiratory system also can limit
performance in patient populations with restricted or obstructed airways. For
example, asthma causes constrtiction of the bronchial tubes and swelling of the
mucous membranes. These effects cause considerable resistance to ventilation,
resulting in a shortness of breath. Exercise is known to bring about symptoms
of asthma, or to worsen those symptoms in select individuals. The mechanism or
mechanisms through which exercise induces airway obstruction in individuals
with so-called exercise-induced asthma remain unknown, despite extensive study.
Respiratory regulation of acid-base balance
As already noted, high-intensity exercise
results in the production and accumulation of lactate and H+.
Although regulation of acid-base balance involves more than control or
respiration, it is discussed here because the respiratory system plays such a
crucial role in rapid adjustment of the body’s acid-base status during and
immediately after exercise.
Acids, such as lactic acid and carbonic acid,
release H+. The metabolism of carbohydrate, fat, or protein produces
inorganic acids that dissociate, increasing the H+ concentration in
body fluids, thus lowering the pH. To minimize the effects of free H+,
the blood and muscles contain base substances that combine with, and thus
buffer or neutralize, the H+:
H+
+ buffer ---> H-buffer
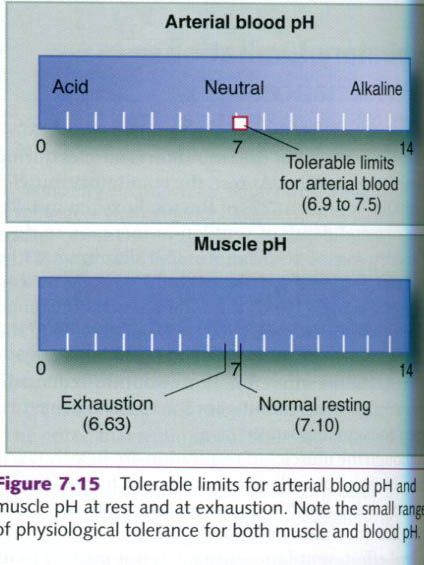
Under resting
conditions, body fluids have more bases(such as bicarbonate, phosphate, and proteins) than acids, resulting in a
slightly alkaline tissue pH that ranges from 7.1 in muscle to 7.4 in arterial
blood. The tolerable limits for arterial blood pH extend from 6.9 to 7.5,
although the extremes of this range can be tolerated only for a few minutes(see
figure below). An H+ concentration above normal(low pH) is referred
to as acidosis, whereas a decrease in H+ below the normal
concentration(high pH) is termed alkalosis.
The pH of
intra- and extracellular body fluids is kept within a relatively narrow range
by:
- Chemical
buffers in the blood,
- Pulmonary
ventilation,
- Kidney
function.
The three major
chemical buffers in the body are bicarbonate(HCO3-),
inorganic phosphates(P), and proteins.
In addition to these, hemoglobin in the red blood cells is also a major buffer.
Table below illustrates the relative contributions of these buffers in handling
acids in the blood. Recall that bicarbonate combines with H+ to form
carbonic acid, thereby eliminating the acidifying influence of free H+.
The carbonic acid in turn forms carbon dioxide and water in the lungs. The CO2
is then exhaled and only water remains.
|
Buffering
capacity of blood components
|
||
|
Buffer
|
Slykes
|
%
|
|
Bicarbonate
|
18.0
|
64
|
|
Hemoglobin
|
8.0
|
29
|
|
Proteins
|
1.7
|
6
|
|
Phosphates
|
0.3
|
1
|
|
Total
|
28.0
|
100
|
The amount of
bicarbonate that combines with H+ equals the amount of acid buffered. When
lactic acid decreases the pH from 7.4 to 7.0, more than 60% of the bicarbonate
initially present in the blood has been used. Even under resting conditions,
the acid produced by the end products of metabolism would use up a major
portion of the bicarbonate from the blood if there were no other way of
removing H+ from the body. Blood and chemical buffers are required
only to transport metabolic acids from their sites of production(the muscles)
to the lungs or kidneys, where they can be removed. Once H+ is
transported and removed, the buffer molecules can be reused.
In the muscle
fibers and the kidney tubules. H+ is primarily buffered by phosphates, such as
phosphoric acid and sodium phosphate. Less is known about the capacity of the
buffers intracellularly, although cells contain more protein and phosphates and
less bicarbonate than do the extracellular fluids.
As noted earlier,
any increase in free H+ in the blood stimulates the respiratory
center to increase ventilation. This facilitates the binding of H+
to bicarbonate and removal of carbon dioxide. The end result is a decrease in
free H+ and in increase in blood pH. Thus, both the chemical buffers
and the respiratory system provide short-term means of neutralizing the acute
effects of exercise acidosis. To maintain a constant buffer reserve, the
accumulated H+ is removed from the body via excretion by the kidneys
and eliminated in urine. The kidneys filter H+ from the blood along with other
waste products. This provides a way to eliminate H+ from the body while
maintaining the concentration of extracellular bicarbonate.
During sprint
exercise, muscle glycolysis generates a large amount of lactate and H+, which
lowers the muscle pH from a resting level of 7.1 to less than 6.7. As shown in
the table below, an all-out 400m sprint decreases leg muscle pH to 6.63 and
increases muscle lactate from a resting value of 1.2 mmol/kg to almost 20
mmol/kg of muscle. Such disturbances in acid-base balance can impair muscle
contractility and its capacity to generate adenosine triphosphate(ATP). Lactate
and H+ accumulate in the muscle, in part because they do not freely
diffuse across the skeletal muscle fiber membranes. Despite the great
production of lactate and H+ during the nearly 60s required to run 400m, these
by-products diffuse throughout the body fluids and reach equilibrium after only
about 5 to 10 min of recovery. Five minutes after the exercise, the runners
described in the table below had blood pH values of 7.10 and blood lactate
concentrations of 12.3 mmol/L, compared with a resting pH of 7.40 and a resting
lactate level of 1.5 mmol/L.
|
Blood
and muscle pH and lactate concentration 5 minutes after a 400m run
|
|||||
|
|
|
Muscle
|
Blood
|
||
|
Runner
|
Time(s)
|
pH
|
Lactate(mmol/kg)
|
pH
|
Lactate(mmol/kg)
|
|
1
|
61.0
|
6.68
|
19.7
|
7.12
|
12.6
|
|
2
|
57.1
|
6.59
|
20.5
|
7.14
|
13.4
|
|
3
|
65.0
|
6.59
|
20.2
|
7.02
|
13.1
|
|
4
|
58.5
|
6.68
|
18.2
|
7.10
|
10.1
|
|
Average
|
60.4
|
6.64
|
19.7
|
7.10
|
12.3
|
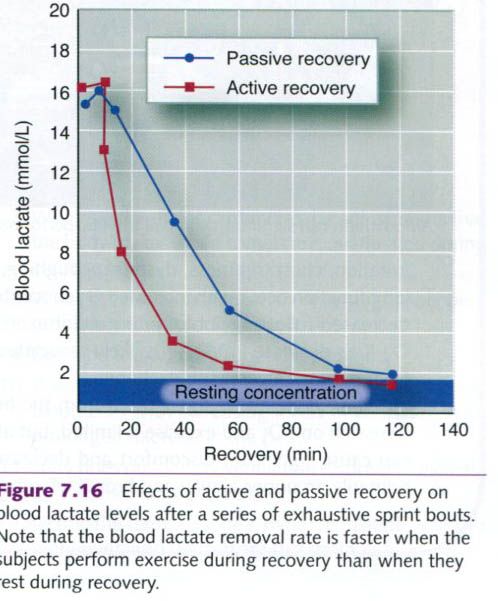
Reestablishing
normal resting concentrations of blood and muscle lactate after such an
exhaustive exercise bout is a relatively slow process, often requiring 1 to 2h.
As shown in the figure below, recovery of blood lactate to the resting level is
facilitated by continued lower-intensity exercise, called active recovery.
After a series of exhaustive sprint bouts, the participants in this study
either sat quietly(passive recovery) or exercised at an intensity of 50% of VO2max.
Blood lactate is removed more quickly during active recovery because the
activity maintains elevated blood flow through the active muscles, which in
turn enhances both lactate diffusion out of the muscles and lactate oxidation.
Although blood
lactate remains elevated for 1 to 2h after highly anaerobic exercise, blood and
muscle H+ concentrations return to normal within 30 to 40 min of
recovery. Chemical buffering, principally by bicarbonate, and respiratory
removal of excess carbon dioxide are responsible for this relatively rapid
return to normal acid-base balance.









0 коментара:
Постави коментар